INDICATIONS AND USAGE
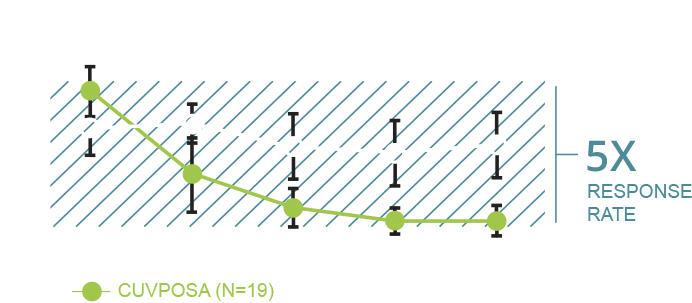
CUVPOSA (glycopyrrolate) oral solution is indicated to reduce chronic severe drooling in patients aged 3-16 years with neurologic conditions associated with problem drooling (e.g., cerebral palsy).
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
CUVPOSA is contraindicated in:
- Patients with medical conditions that preclude anticholinergic therapy (e.g., glaucoma, paralytic ileus, unstable cardiovascular status in acute hemorrhage, severe ulcerative colitis, toxic megacolon complicating ulcerative colitis, myasthenia gravis).
- Patients taking solid oral dosage forms of potassium chloride tablets through the gastrointestinal (GI) tract may be arrested or delayed with coadministration of CUVPOSA.
WARNINGS AND PRECAUTIONS
- Constipation is a common dose-limiting adverse reaction which sometimes leads to glycopyrrolate discontinuation. Assess patients for constipation, particularly within 4-5 days of initial dosing or after a dose increase. Intestinal pseudo-obstruction has been reported and may present as abdominal distention, pain, nausea, or vomiting.
- Diarrhea may be an early symptom of incomplete mechanical intestinal obstruction, especially in patients with ileostomy or colostomy. If incomplete mechanical intestinal obstruction is suspected, discontinue treatment with CUVPOSA and evaluate for intestinal obstruction.
- In the presence of high ambient temperature, heat prostration (fever and heat stroke due to decreased sweating) can occur with the use of anticholinergic drugs such as CUVPOSA. Advise patients/caregivers to avoid exposure of the patient to hot or very warm environmental temperatures.
- CUVPOSA may produce drowsiness or blurred vision. As appropriate for a given age, warn the patient not to engage in activities requiring mental alertness such as operating a motor vehicle or other machinery, or performing hazardous work while taking CUVPOSA.
- Use CUVPOSA with caution in patients with conditions that are exacerbated by anticholinergic drug effects including:
-
- Autonomic neuropathy
- Renal disease
- Ulcerative colitis – Large doses may suppress intestinal motility to the point of producing a paralytic ileus and for this reason may precipitate or aggravate “toxic megacolon”, a serious complication of the disease
- Hyperthyroidism
- Coronary heart disease, congestive heart failure, cardiac tachyarrhythmia, tachycardia, and hypertension
- Hiatal hernia associated with reflux esophagitis, since anticholinergic drugs may aggravate this condition
ADVERSE REACTIONS
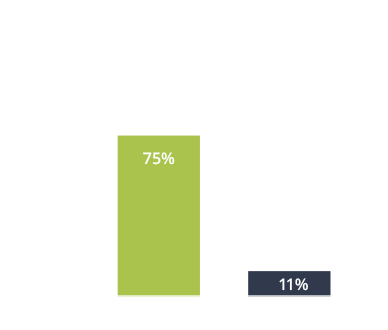
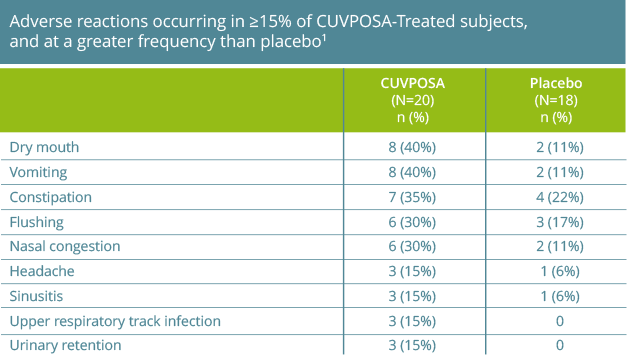
The most common adverse reactions reported with CUVPOSA are dry mouth, vomiting, constipation, flushing, and nasal congestion.
The most commonly observed adverse reactions reported by ≥15% of CUVPOSA-treated patients for the placebo-controlled clinical trial were:
- dry mouth (40%)
- vomiting (40%)
- constipation (35%)
- flushing (30%)
- nasal congestion (30%)
- headache (15%)
- sinusitis (15%)
- upper respiratory tract infection (15%)
- urinary retention (15%)
The most commonly observed adverse reactions which occurred at a rate of <2% of CUVPOSA-treated patients in the open label study were:
- Gastrointestinal: abdominal distension, abdominal pain, stomach discomfort, chapped lips, flatulence, retching, dry tongue
- General Disorders: irritability, pain
- Infections: pneumonia, sinusitis, tracheostomy infection, upper respiratory tract infection, urinary tract infection
- Investigations: heart rate increase
- Metabolism and Nutrition: dehydration
- Nervous System: headache, convulsion, dysgeusia, nystagmus
- Psychiatric: agitation, restlessness, abnormal behavior, aggression, crying, impulse control disorder, moaning, mood altered
- Respiratory: increased viscosity of bronchial secretion, nasal congestion, nasal dryness
- Skin: dry skin, pruritus, rash
- Vascular: pallor
Additional adverse reactions identified during post approval use of glycopyrrolate tablets include: loss of taste and suppression of lactation.
DRUG INTERACTIONS
Drugs Affected by Reduced GI Transit Time
Glycopyrrolate reduces GI transit time, which may result in altered release of certain drugs when formulated in delayed- or controlled-release dosage forms.
- The passage of potassium chloride tablets through the GI tract may be arrested or delayed with coadministration of glycopyrrolate. Solid dosage forms of potassium chloride are contraindicated.
- Digoxin administered as slow dissolution oral tablets may have increased serum levels and enhanced action when administered with glycopyrrolate. Monitor patients receiving slow dissolution digoxin for increased action if glycopyrrolate is administered regularly. Consider the use of other oral dosage forms of digoxin (e.g., elixir or capsules).
Amantadine
The anticholinergic effects of glycopyrrolate may be increased with concomitant administration of amantadine. Consider decreasing the dose of glycopyrrolate during coadministration of amantadine.
Drugs Whose Plasma Levels May be Increased by Glycopyrrolate
Coadministration of glycopyrrolate may result in increased levels of certain drugs.
- Atenolol's bioavailability may be increased with coadministration of glycopyrrolate. A reduction in the atenolol dose may be needed.
- Metformin plasma levels may be elevated with coadministration of glycopyrrolate, increasing metformin's pharmacologic and toxic effects. Monitor clinical response to metformin with concomitant glycopyrrolate administration; consider a dose reduction of metformin if warranted.
Drugs Whose Plasma Levels May be Decreased by Glycopyrrolate
Coadministration of glycopyrrolate may result in decreased levels of certain drugs.
- Haloperidol's serum level may be decreased when coadministered with glycopyrrolate, resulting in worsening of schizophrenic symptoms, and development of tardive dyskinesia. Closely monitor patients if coadministration cannot be avoided.
- Levodopa's therapeutic effect may be reduced with glycopyrrolate administration. Consider increasing the dose of levodopa.
USE IN PREGNANCY
There are no available data in pregnant women for CUVPOSA to inform decisions concerning any drug-associated risks.
PEDIATRIC USE
CUVPOSA was evaluated for chronic severe drooling in patients aged 3-16 years with neurologic conditions associated with problem drooling. CUVPOSA has not been studied in subjects under the age of 3 years.
Please see full Prescribing Information.